Pathologic activation of the phosphatidylinositol 3-kinase (PI3K)/Akt signaling pathway is an oncogenic driver for many malignancies, including lymphomas1. Although validated as a therapeutic oncologic target, the PI3K signaling pathway is also implicated in normal glucose homeostasis. Specifically, since the PI3K subunit p110α is chiefly responsible for downstream insulin receptor (INSR) signaling, PI3K signaling inhibition that includes p110α leads to severe hyperglycemia. Both compensatory endogenous insulin release as well as treatment of hyperglycemia with exogenous insulin/insulin mimetics activate INSR-associated PI3K signaling, ultimately limiting blockade of tumor associated PI3K signaling2. Aggressive lymphomas driven by pathologic PI3K signaling respond to combined PI3Kα and δ/γ inhibition, but this is likely submaximal due to endogenous insulin feedback, and more intense dosing is difficult to achieve safely due to severe hyperglycemia3. Therefore, a novel strategy to maintain blockade of tumor associated PI3K signaling while reducing hyperglycemia is needed. We hypothesized that inhibition of glucagon receptor (GCGR) signaling, a pathway that does not depend on PI3K, may normalize PI3K inhibitor induced hyperglycemia without disrupting antitumor PI3K blockade. GCGR blockade with a GCGR specific monoclonal antibody (REMD-477, a human anti-GCGR antibody, or REMD2.59c, a murine equivalent) may allow for the safer use of more potent PI3K inhibitors or more intensive dosing schedules in the treatment of aggressive lymphomas.
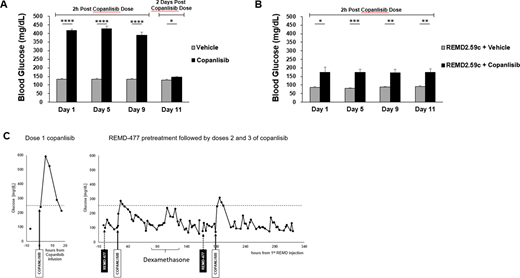
The effect of GCGR blockade on PI3K inhibitor induced hyperglycemia was first evaluated in a non-tumor bearing mouse model using REMD2.59c. The PI3K inhibitor copanlisib was administered by tail vein injection to CB17 SCID mice in the control group every other day at the reported MTD of 15 mg/kg. REMD2.59c was administered by intraperitoneal injection to the treatment group intermittently on the day prior to copanlisib injection. Blood glucose levels were measured 2 hours after copanlisib injection. Significant hyperglycemia was observed after copanlisib treatment, and this was corrected by REMD2.59c pretreatment, confirming the effectiveness of GCGR blockade (Figure A-B).
Having demonstrated that GCGR blockade controls hyperglycemia caused by PI3K inhibition with copanlisib in a mouse model, we next treated a 54 year old non-diabetic woman with relapsed and refractory peripheral T cell lymphoma (PTCL) on an IRB-approved clinical pilot study of copanlisib in combination with REMD-477. Following copanlisib 60mg IV alone, glucose increased from a baseline of 100mg/dl to a maximum of 592 mg/dl at 5 hours. For the following weekly doses of copanlisib, she was pre-treated with 70mg REMD-477 SQ, and copanlisib induced hyperglycemia was significantly ameliorated (Figure C). The patient tolerated treatment with REMD-477 well without significant episodes of hypoglycemia.
With improved glycemic control on REMD-477, our patient did not require dose reductions or treatment delays, and experienced clinical improvement in lymphadenopathy on copanlisib. She had previously progressed through 6 lines of therapy, including duvelisib, a selective PI3K δ/γ inhibitor which necessitated dose reduction due to gastrointestinal toxicity. Our study of the GCGR mAb REMD-477 as a novel strategy to counteract PI3K inhibitor induced hyperglycemia and insulin feedback is an important advance that may allow for more effective and safer use of potent PI3K inhibitors in the treatment of aggressive lymphomas.
References
1. Alzahrani AS. PI3K/Akt/mTOR inhibitors in cancer: At the bench and bedside. Semin Cancer Biol. 2019;59(April):125-132. doi:10.1016/j.semcancer.2019.07.009
2. Hopkins BD, Pauli C, Du X, et al. Suppression of insulin feedback enhances the efficacy of PI3K inhibitors. Nature. 2018;560(7719):499-503. doi:10.1016/j.physbeh.2017.03.040
3. Cheson BD, O'Brien S, Ewer MS, et al. Optimal Management of Adverse Events From Copanlisib in the Treatment of Patients With Non-Hodgkin Lymphomas. Clin Lymphoma, Myeloma Leuk. 2019;19(3):135-141. doi:10.1016/j.clml.2018.11.021
Calzone:REMD Biotherapeutics: Current equity holder in private company. Yan:REMD Biotherapeutics: Current Employment, Current equity holder in private company. Thai:REMD Biotherapeutics: Current Employment, Current equity holder in private company. Lyerly:REMD Biotherapeutics: Consultancy, Current equity holder in private company.
Copanlisib in the treatment of peripheral T cell lymphoma
Author notes
Asterisk with author names denotes non-ASH members.